Toxoplasma gondii is a single-cell parasite that infects mammals and birds. On average, one third of human population is chronically infected. Infection of T. gondii can cause congenital, ocular or acute disseminated diseases in healthy individuals, and severe encephalitis in immunocompromised individuals such as AIDS patients.
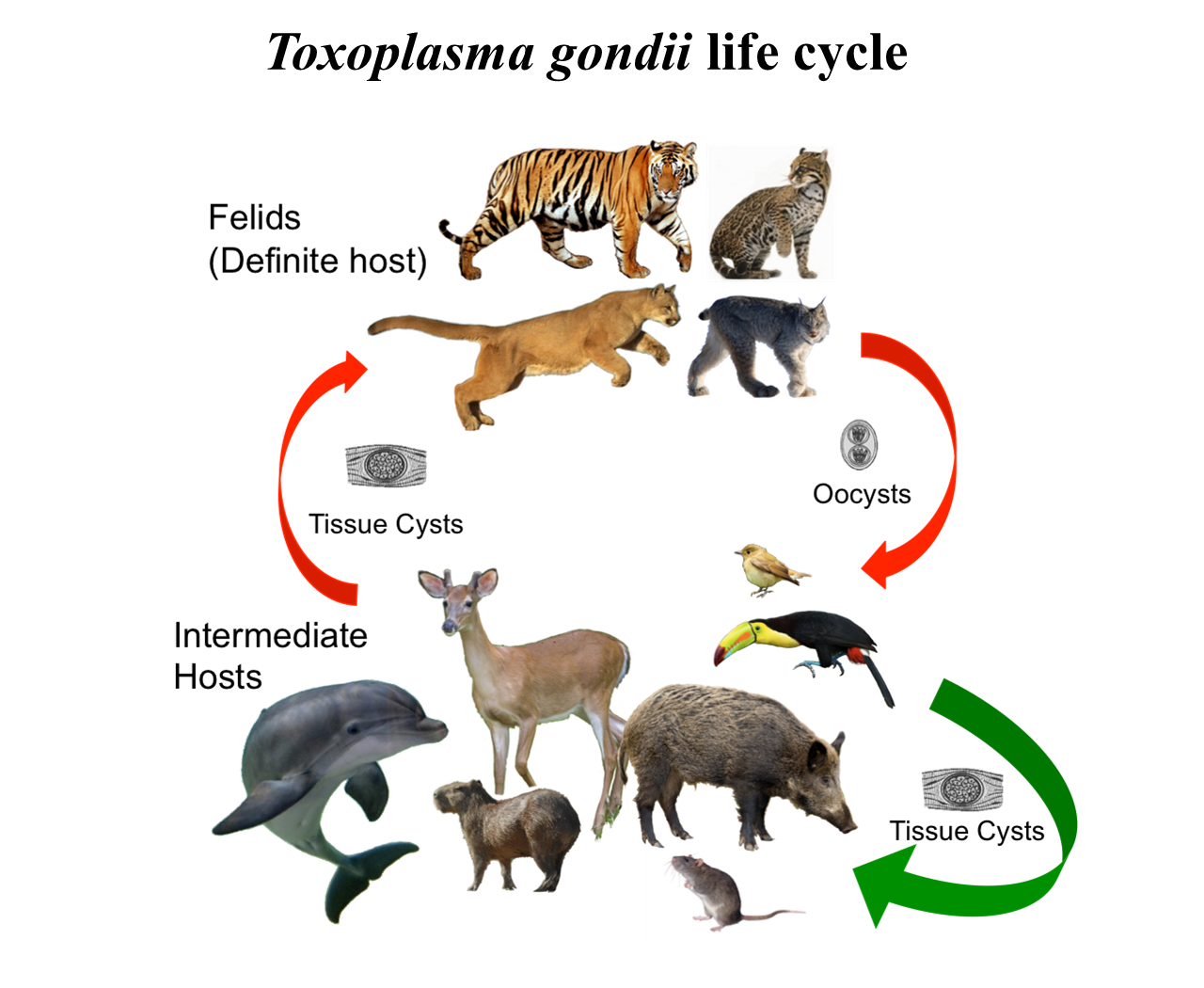
Toxoplasma gondii has a complex life cycle. Felids (domestic and wild cats) are the only known definitive hosts, in which the parasite can reproduce sexually to generate hundreds of millions of oocysts that are shed into the environment in cat feces. Within a few days, oocysts in the environment mature to produce infectious parasites called sporozoites. All warm-blooded vertebrates (mammals and birds) can serve as intermediate hosts, which acquire infection through ingesting soil, water, or plants contaminated with oocysts. Parasites in an intermediate host undergo asexual replication. In about a week, the parasites may switch to a slow-growing phase (bradyzoites), producing relatively quiescent tissue cysts, which are often localized in brain and muscle tissues. Transmission also occurs through cannibalism, predation and scavenging among intermediate hosts. Humans become infected with T. gondii congenitally or by ingestion of either oocysts in contaminated food and water, or tissue cysts in undercooked meat of infected animals. Upon ingestion of oocysts or tissue cysts, the parasites are released, penetrate the intestinal cells, and replicate rapidly inside host cells. Rapid replication is responsible for systemic dissemination of the parasite, and most of the pathology associated with acute toxoplasmosis. Host immune response eventually eliminates fast-replicating parasites but a population of parasites switch to slow-replicating stage that encyst and establish chronic infection primarily in the brain and muscle tissue. Severe disease results in immune suppressed individuals (e.g., AIDS patients) due to reactivation of parasite replication in the absence of effective immune control. Currently there are no therapeutics capable of eliminating the chronic stage of T. gondii infection.
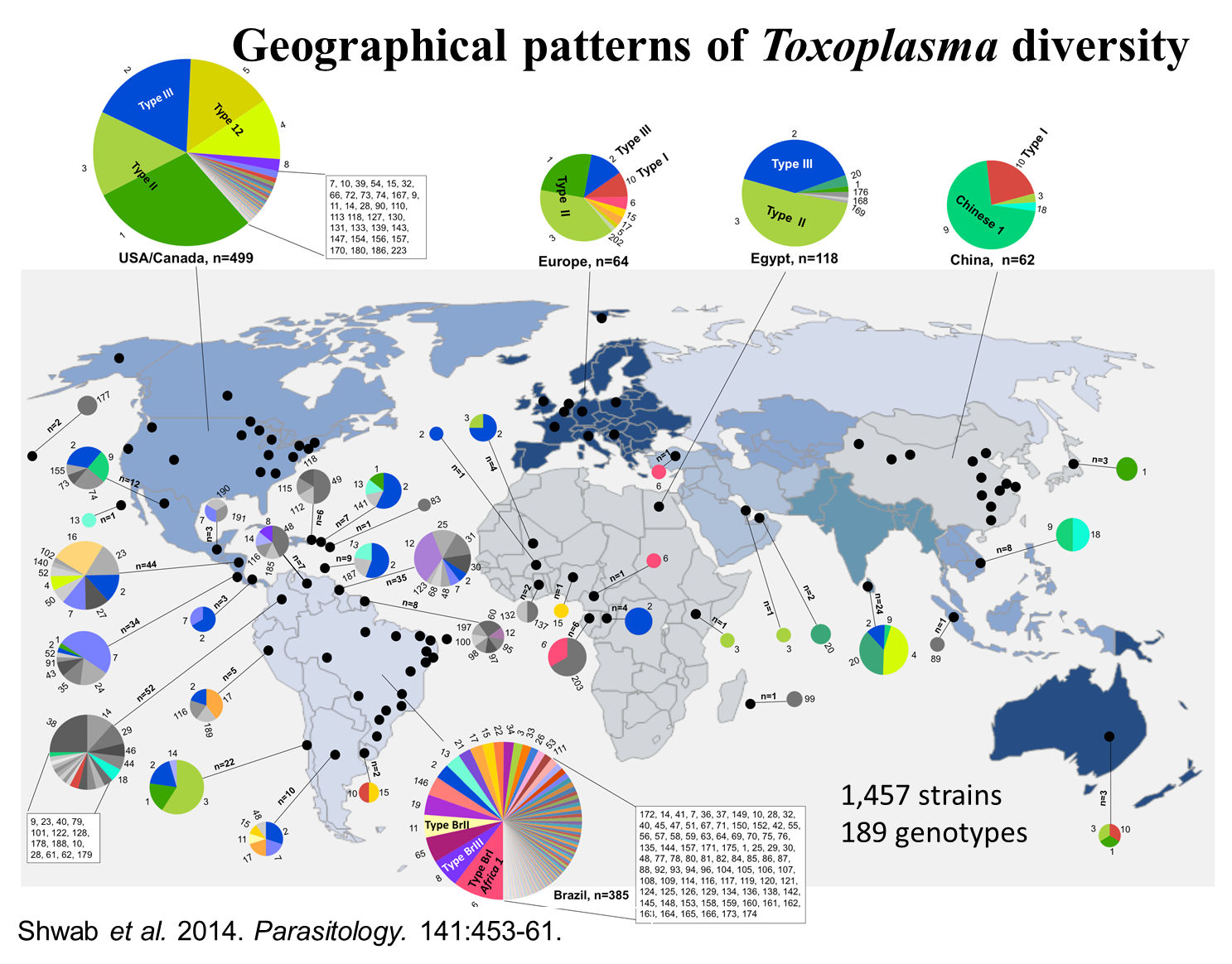
Our laboratory is interested in studying molecular epidemiology, population genetics and evolution of T. gondii. Our goal is to reveal major reservoirs and transmission modes through which the parasite spreads among different hosts, particularly in humans. To reach this goal, we first developed a simple and cost-effective molecular method to identify T. gondii isolates (Su et al., Int. J. Parasitol. 2006, 36:841-848; Su et al., Parasitology, 2010; 137:1-11), and applied the method to thousands of samples collected worldwide. Our studies revealed global patterns of genetic diversity of T. gondii (Su et al., PNAS, 2012, 109:5844-5849; Shwab et al., Parasitology, 2014, 141:453-461). Our analysis revealed distinct distribution patterns of T. gondii types for each major geographic region examined, with striking contrast between the highly diverse of the Central/South American region and the more clonal populations found in all other areas. North America, Europe, North Africa, and East Asia are each dominated by particular clonal types. Interestingly, T. gondii strains from Central/South America were more likely to be virulent. These results set the stage for future studies aimed to identify the factors that may drive population diversity and the factors responsible for rapid expansion of this parasite on our planet.
If you need MAT antigens for research, please visit website UT Research Foundation. For detailed information of the antigens, check the link here.
Dr. Chunlei Su
Hesler Biology Building, 402A
Department of Microbiology
University of Tennessee
Knoxville, TN 37996
Email: csu1@utk.edu